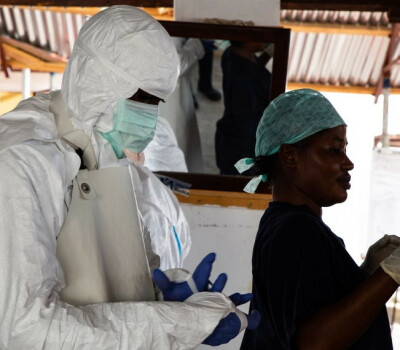
Last study participant followed up in unique Ebola vaccine booster trial in DRC

On Friday 17 April, the final participant in the EBO-BOOST clinical trial attended a routine follow-up visit at the Laboratoire provincial de santé publique et recherche in Mbandaka, the capital of Equateur Province in the Democratic Republic of the Congo (DRC). This moment marks an important milestone in the research on the optimal combination of Ebola booster vaccines and how the immune system responds to different combinations of two licensed Ebola vaccines.
Header image: Study doctor Jessy Sabue (INRB) explains the study procedure to participants at the Provincial Laboratory in Mbandaka, Equateur Province, Democratic Republic of the Congo.

Towards flexible Ebola vaccination strategies
EBO-BOOST is a clinical trial investigating how Ebola booster vaccines can best be deployed. Although two Ebola vaccine regimens are currently approved, it remains unclear how long protection lasts and what the optimal revaccination strategy is. The study compares different combinations in people who were previously vaccinated and examines whether combining different vaccines ("mix-and-match") is as safe and effective as repeating the same vaccine. Through this project, researchers aim to accelerate vaccine deployment during outbreaks, develop clear revaccination guidelines, and strengthen local epidemic preparedness.
"With this trial, we want to create more flexibility in vaccination strategies," says Prof Wim Adriaensen, Head of the Clinical Immunology Unit at the Institute of Tropical Medicine and principal investigator of the study. "If different vaccines prove to be interchangeable, we can respond more quickly and efficiently to outbreaks, depending on local conditions and vaccine availability."
Ebola remains a threat
Since the discovery of the virus in 1976, multiple outbreaks have occurred, especially in countries in Central and West Africa, including the DRC. The disease causes severe symptoms such as high fever, vomiting, and internal bleeding, and has a high mortality rate.
Mbandaka, the capital of Equateur Province with more than one million inhabitants, has experienced several outbreaks in recent years, including as recently as 2022. Thanks to international efforts, two vaccine regimens (by MSD and J&J) have been developed and licensed, but the duration of that protection remains unknown. This stresses the need for booster doses, but evidence-based revaccination guidelines are lacking. The EBO-BOOST project aims to address this gap.
"It is quite unique to mix the two vaccines in Ebola revaccination strategies in the DRC,” says Prof Hugo Kavunga-Membo, principal investigator at INRB, the Congolese National Institute of Biomedical Research. “Studying the best strategy to achieve the strongest and longest lasting response after revaccination have never been studied before. That makes this study truly innovative, in particular for our country that regularly faces epidemics.”
Final study participant followed up
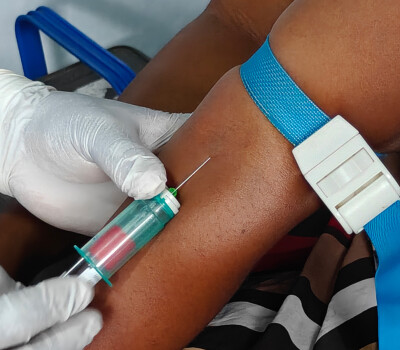
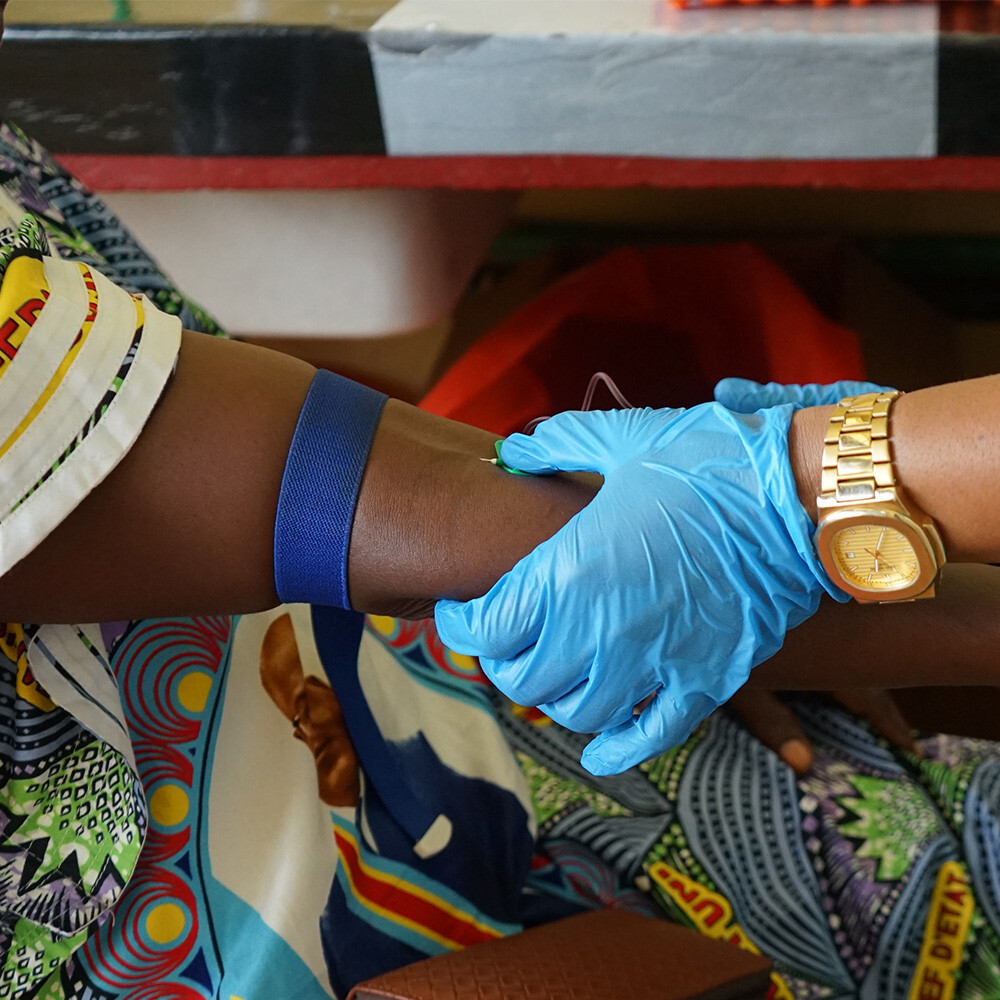
The last group of study participants visited the provincial laboratory in Mbandaka during the week of 13 April for a routine check-up, blood sampling, and an oral swab, six months after receiving a booster vaccine.
For the study, participants were recruited who had received an Ebola vaccine one to seven years earlier. As it is not yet fully understood how protective immunity works, the researchers are analysing both antibody and immune cell levels in a holistic manner to fully understand how strong the immune response is and how long it lasts.

Next steps
In the coming months, the collected samples will be further analysed in the INRB laboratory in Kinshasa. In addition to measuring the quality of the antibodies to neutralise the virus, researchers will also thaw the frozen immune cells (which remain in the body long-term as memory cells) and map their response to Ebola using new tools now available in the laboratory.
The impact of EBO-BOOST goes beyond the results alone. Thanks to investments in local research infrastructure, complex immunological analyses can now be carried out within the DRC, without the need to send samples abroad. This strengthens preparedness for future epidemics—not only Ebola, but also other infectious diseases.
The study is a strong collaboration with the Institut National de Recherche Biomédicale (INRB), a long-standing ITM partner. It is funded by the Coalition for Epidemic Preparedness Innovations (CEPI), Merck Sharp & Dohme (MSD), Johnson & Johnson, and the Directorate-General for Development Cooperation and Humanitarian Aid (DGD).

EBO-BOOST
We aim to address the ongoing threat of the Ebola Virus Disease by investigating optimal timing and safety of booster vaccinations for rapid deployment during outbreaks, and strengthening local epidemic preparedness.
Spread the word! Share this story on