Institute of Tropical Medicine
Global Science for Health Worldwide
Tropical diseases, HIV/AIDS, tuberculosis and inadequate health care affect the lives of billions of people worldwide.
Established in 1906, the Institute of Tropical Medicine (ITM) in Antwerp (Belgium) strives for the advancement of science and health for all, through innovative research, advanced education, professional medical services and capacity sharing with partner institutions in Africa, Asia and Latin America. We aim to reduce the impact of diseases and health issues on populations globally.
For us, scientific excellence and societal impact are two sides of the same coin.
Highlights

Read our annual report
The Institute was thriving in 2024. Discover our highlights in our newly published annual report.

Win cinema tickets!
Did you recently have an appointment at our polyclinic? Share your opinion and get a chance to win two cinema tickets!

Podcast season 3 out now!
Listen to The Road to Zero, the third season of our award-winning podcast Transmission, on your favourite podcast platform! 🎧

Our clinic
We are an outpatient clinic specialised in the diagnosis, treatment and care of HIV/AIDS and other sexually transmitted infections, travel health and tropical medicine, and infectious diseases.
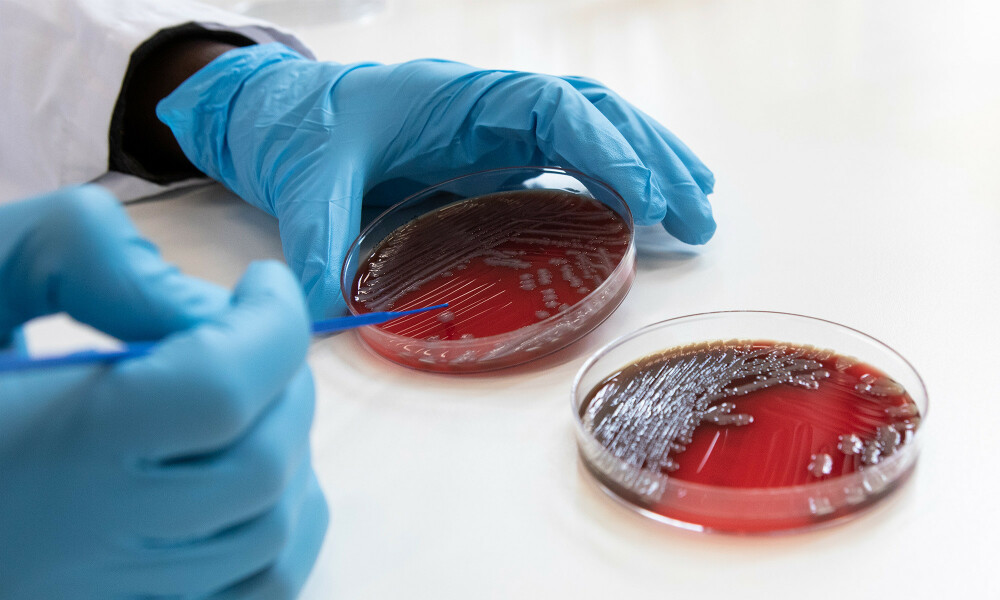
Our research
We aim to advance knowledge about tropical and infectious diseases and tackle important health challenges, with a particular focus on low-resource settings and vulnerable populations.
Our courses
We offer master's programmes, postgraduate certificate programmes and expert short courses in tropical medicine, international public health and global One Health for professionals from around the world.

Our partnerships
We work with numerous partners across three continents on institutional capacity sharing and collaboration.

Make an appointment
Consultations in our polyclinic are by appointment only. Book your appointment well in advance, as waiting times can be long in spring and summer.

Contact
You need to make an appointment at our clinic to receive medical advice, but there are a few other ways you can get in touch.
Webshop
Explore our webshop and get your hands on an ITM hoodie, notebook, pair of socks, boc 'n roll, and so much more!
All events
Search eventsITM listens, acts and commits at Antwerp Pride 2025!
Pride Village, Cockerillkaai, Antwerpen
Open Monuments Day 2025
Instituut voor Tropische Geneeskunde (ITG)
International Course on Principles of Biosafety 2025
UAntwerp and ITM